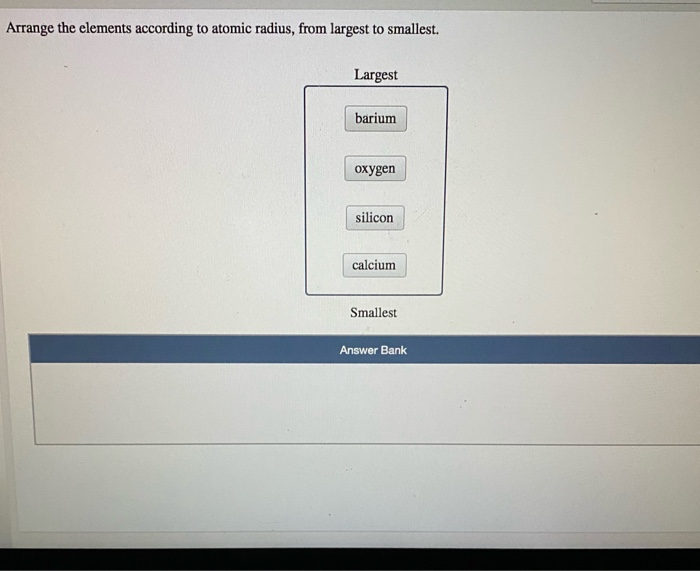
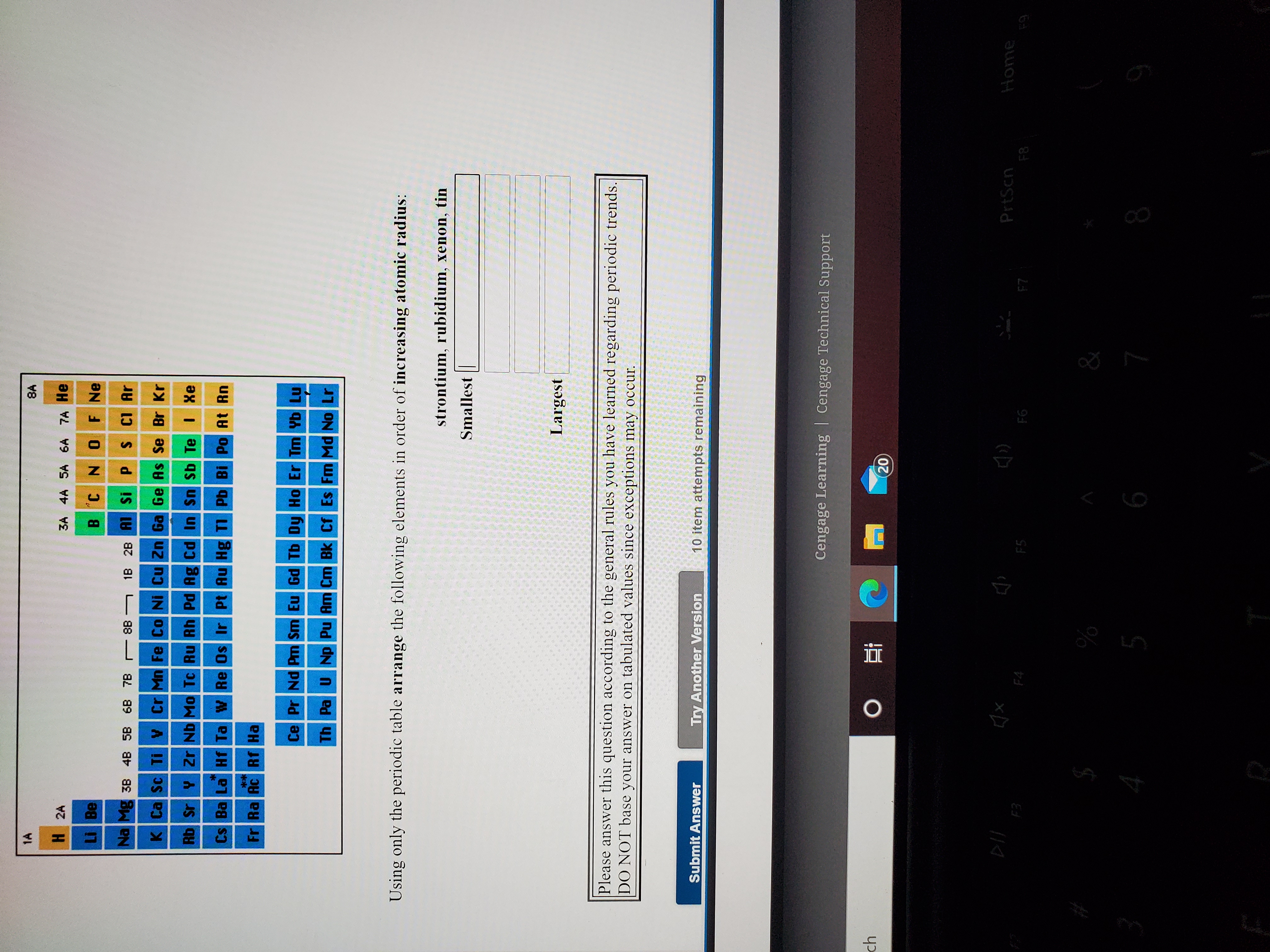
> C, O and F are elements of the same period and in the same period atomic. Why does radius increase with higher atomic numbers in a group? As you move down a group in the periodic table, additional layers of electrons are being added, which naturally causes the ionic radius to increase as you move down the periodic table. So, its atomic radius will be more than second period elements and less than bromine. While the atomic radius follows a similar trend, ions may be larger or smaller than neutral atoms.For example, argon (Ar, atomic mass 39.9) is placed before potassium (K, atomic mass 39.1). In these cases, Mendeleev placed elements according to similarities in their properties and not in increasing order of their atomic masses. We store cookies data for a seamless user experience. Largest Smallest Answer Bank oxygen silicon calcium barium Arrange these elements according to atomic radius. Rank these atoms and ions according to radius from smallest to largest. Anomalous pairs: In certain pairs of elements, the increasing order of atomic masses was not obeyed. Largest radius Smallest radius Answer Bank Ar Si Mg Al Na Arrange the elements according to atomic radius, from largest to smallest. Arrange the following elements according to atomic radius: S, Ar, Si, Mg, Cl, Al, Na, and P. But for the nonmetallic elements, the ionic radius increases because there are more electrons than protons. Rank these elements according to first ionization energy from highest to lowest. Arrange the following elements according to atomic radius: S, Ar, Si, Mg, Cl, Al, Na, and P. More protons are added, but the outer valence shell remains the same, so the positively charged nucleus draws in the electrons more tightly. 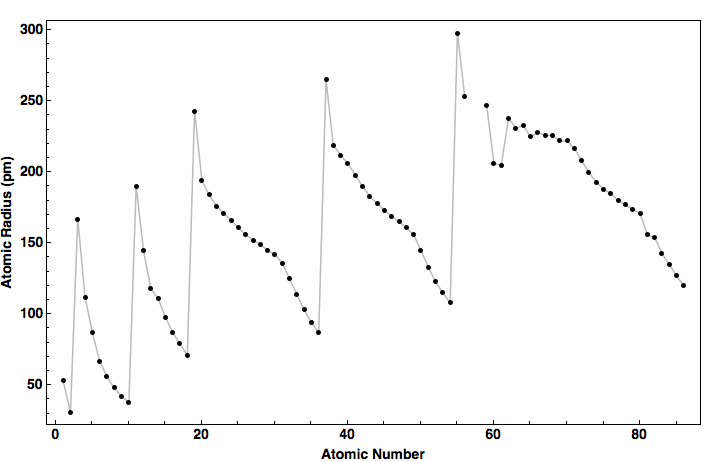
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
